 
They form the exterior of an atom and specify the rules of chemistry by which atoms combine to form molecules.Įlectrons are in continuous motion around the nucleus, but motions on this submicroscopic scale obey different laws from those we are familiar with in everyday life. 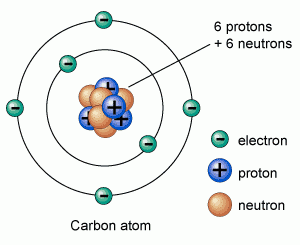
In living tissues, it is only the electrons of an atom that undergo rearrangements. Protons and neutrons are welded tightly to one another in the nucleus and change partners only under extreme conditions-during radioactive decay, for example, or in the interior of the sun or of a nuclear reactor. To understand how atoms bond together to form the molecules that make up living organisms, we have to pay special attention to their electrons. The Outermost Electrons Determine How Atoms Interact This quantity is called one mole of the substance ( Figure 2-2). If a substance has a molecular weight of X, 6 × 10 23 molecules of it will have a mass of X grams. This huge number (6 × 10 23, called Avogadro's number) is the key scale factor describing the relationship between everyday quantities and quantities measured in terms of individual atoms or molecules. One proton or neutron weighs approximately 1/(6 × 10 23) gram, so one gram of hydrogen contains 6 × 10 23 atoms. An individual carbon atom is roughly 0.2 nm in diameter, so that it would take about 5 million of them, laid out in a straight line, to span a millimeter. The mass of an atom or a molecule is often specified in daltons, one dalton being an atomic mass unit approximately equal to the mass of a hydrogen atom.Ītoms are so small that it is hard to imagine their size. Thus the major isotope of carbon has an atomic weight of 12 and is symbolized as 12C, whereas the unstable isotope just discussed has an atomic weight of 14 and is written as 14C. This is essentially equal to the number of protons plus neutrons that the atom or molecule contains, since the electrons are much lighter and contribute almost nothing to the total. The atomic weight of an atom, or the molecular weight of a molecule, is its mass relative to that of a hydrogen atom. This forms the basis for a technique known as carbon 14 dating, which is used in archaeology to determine the time of origin of organic materials. Carbon 14 undergoes radioactive decay at a slow but steady rate. For example, while most carbon on Earth exists as the stable isotope carbon 12, with six protons and six neutrons, there are also small amounts of an unstable isotope, the radioactive carbon 14, whose atoms have six protons and eight neutrons. Multiple isotopes of almost all the elements occur naturally, including some that are unstable. Thus an element can exist in several physically distinguishable but chemically identical forms, called isotopes, each isotope having a different number of neutrons but the same number of protons. 
They contribute to the structural stability of the nucleus-if there are too many or too few, the nucleus may disintegrate by radioactive decay-but they do not alter the chemical properties of the atom. Neutrons are uncharged subatomic particles of essentially the same mass as protons. Although the electrons are shown here as individual particles, in reality their behavior is governed by the laws of quantum mechanics, and there is no way of predicting exactly (more.) Highly schematic representations of an atom of carbon and an atom of hydrogen. It is these electrons that determine the chemical behavior of an atom, and all of the atoms of a given element have the same atomic number. Since an atom as a whole is electrically neutral, the number of negatively charged electrons surrounding the nucleus is equal to the number of positively charged protons that the nucleus contains thus the number of electrons in an atom also equals the atomic number. The electric charge carried by each proton is exactly equal and opposite to the charge carried by a single electron. An atom of carbon has six protons in its nucleus and an atomic number of 6 ( Figure 2-1). An atom of hydrogen has a nucleus composed of a single proton so hydrogen, with an atomic number of 1, is the lightest element. The number of protons in the atomic nucleus gives the atomic number. The nucleus in turn consists of two kinds of subatomic particles: protons, which are positively charged, and neutrons, which are electrically neutral. Each atom has at its center a positively charged nucleus, which is surrounded at some distance by a cloud of negatively charged electrons, held in a series of orbitals by electrostatic attraction to the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
